Anavex Life Sciences Reports Fiscal Second Quarter 2018 Financial Results
- ANAVEX® Life Sciences Corp.
- May 9, 2018
- 4 min read
Updated: Jul 11, 2024
Conference Call and Webcast Today at 4:30 p.m. ET

NEW YORK – May 10, 2018
Anavex Life Sciences Corp. (“Anavex” or the “Company”) (Nasdaq: AVXL), a clinical-stage biopharmaceutical company developing differentiated therapeutics for the treatment of neurodegenerative and neurodevelopmental diseases including Alzheimer’s disease, Rett syndrome and other central nervous system (CNS) diseases, today reported financial results for the second fiscal quarter ended March 31, 2018.
Clinical Update:
The Phase 2a Alzheimer’s extension study ANAVEX®2-73-003 has been approved by the Ethics Committee in Australia to continue for a further 104 weeks, i.e. additional two (2) years per request by patients, their caregivers and physicians. The ANAVEX®2-73-003 extension study was the continuation of the 57-week ANAVEX®2-73-002 study and is designed to allow participants who complete the first 104 week extension to continue taking ANAVEX®2-73 for an additional 104 weeks, providing an opportunity to gather extended five (5) years of cumulative safety and tolerability data for orally daily dosed ANAVEX®2-73. The further extension is in addition and independent of the Company’s planned larger Phase 2/3 double-blinded, placebo-controlled study of ANAVEX®2-73 in Alzheimer’s disease.
Financial Highlights:
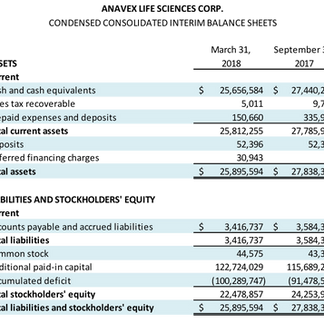
Cash and equivalents of $25.7 million at March 31, 2018, compared to $27.4 million at September 30, 2017.
Cash used to fund operations in the second fiscal quarter was $2.8 million, compared to $0.7 million for the comparative quarter last year. This is in line with the Company’s cash utilization guidance.
Operating expenses in the second fiscal quarter of $4.7 million compared to $3.6 million for the comparative quarter last year. This increase was a result of increased spending on research and development, related to clinical trial preparatory activities.
Net loss for the second fiscal quarter of $4.8 million, or $0.11 per share, compared to a net loss of $1.5 million, or $0.04 per share for the comparative quarter.
“We are looking forward, focusing on executing clinical trials using the inclusion of advanced genomic biomarkers into late-stage CNS precision medicine trials, including our Rett syndrome, Alzheimer’s disease and Parkinson’s disease dementia trials – all indications with high unmet need,” said, Christopher U. Missling, PhD, President and Chief Executive Officer of Anavex.
Recent Corporate Highlights:
April 17, 2018 – Anavex announced that the Company had submitted to regulatory authorities in Europe a Phase 2 Parkinson’s disease dementia study and, pending approval, the Company plans to initiate this clinical trial in calendar H2 2018.
March 7, 2018 – Anavex announced new data for ANAVEX®2-73 in a genetic mouse model of Tuberous Sclerosis Complex (TSC), a rare genetic disorder characterized by the growth of numerous benign tumors in many parts of the body with a high incidence of seizures. The new preclinical data demonstrate that treatment with ANAVEX®2-73 significantly increases survival and reduces seizures.
March 5, 2018 –Stephan Toutain, MS, MBA joined Anavex as Senior Vice President Operations and Claus van der Velden, PhD was appointed to the Board of Directors.
The financial information for fiscal quarter ended March 31, 2018, should be read in conjunction with the Company’s condensed consolidated interim financial statements, which will appear on EDGAR and will be available on the Anavex website at
www.anavex.com.
Conference Call / Webcast Information
Anavex will host a conference call at 4:30 p.m. ET today, May 10, 2018.
The live webcast of the conference call can be accessed online at http://www.wsw.com/webcast/cc/avxl5. To listen to the live call by phone, interested parties within the U.S. should dial, toll-free, 1 (866) 939-3921 and international callers should dial 1 (678) 302-3550. Please use confirmation number 46931087. A replay of the call will be available approximately one hour after the end of the call at www.anavex.com.
About Anavex Life Sciences Corp.
Anavex Life Sciences Corp. (Nasdaq: AVXL) is a publicly traded biopharmaceutical company dedicated to the development of differentiated therapeutics for the treatment of neurodegenerative and neurodevelopmental diseases including Alzheimer’s disease, other central nervous system (CNS) diseases, pain and various types of cancer. Anavex’s lead drug candidate, ANAVEX®2-73, recently completed a successful Phase 2a clinical trial for Alzheimer’s disease. ANAVEX®2-73 is an orally available drug candidate that restores cellular homeostasis by targeting sigma-1 and muscarinic receptors. Preclinical studies demonstrated its potential to halt and/or reverse the course of Alzheimer’s disease. ANAVEX®2-73 also exhibited anticonvulsant, anti-amnesic, neuroprotective and anti-depressant properties in animal models, indicating its potential to treat additional CNS disorders, including epilepsy. The Michael J. Fox Foundation for Parkinson’s Research previously awarded Anavex a research grant to develop ANAVEX®2-73 for the treatment of Parkinson’s disease. The grant fully funded a preclinical study, which could justify moving ANAVEX®2-73 into a Parkinson’s disease clinical trial. ANAVEX®3-71, which targets sigma-1 and M1 muscarinic receptors, is a promising preclinical drug candidate demonstrating disease-modifying activity against the major hallmarks of Alzheimer’s disease in transgenic (3xTg-AD) mice, including cognitive deficits, amyloid and tau pathologies. In preclinical trials, ANAVEX®3-71 has shown beneficial effects on neuroinflammation and mitochondrial dysfunction. Further information is available at www.anavex.com. You can also connect with the company on Twitter, Facebook and LinkedIn.
Forward-Looking Statements
Statements in this press release that are not strictly historical in nature are forward-looking statements. These statements are only predictions based on current information and expectations and involve a number of risks and uncertainties. Actual events or results may differ materially from those projected in any of such statements due to various factors, including the risks set forth in the Company’s most recent Annual Report on Form 10-K filed with the SEC. Readers are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof. All forward-looking statements are qualified in their entirety by this cautionary statement and Anavex Life Sciences Corp. undertakes no obligation to revise or update this press release to reflect events or circumstances after the date hereof.
For Further Information:
Anavex Life Sciences Corp. Research & Business Development Toll-free: 1-844-689-3939 Email: info@anavex.com
Investors & Media:
Meggie Purcell Solebury Trout mpurcell@soleburytrout.com